TRAINING DAYS: 5
(4 days of training + 1 day exam)
CPD CERTIFICATION
31 Credits
EXAM DURATION
3 Hours
EXAM RETAKE POSSIBLE?
Yes. Free one more exam in 12 months
WHAT IS INCLUDED?
Training, PECB exam and certification
Why Should You Attend?
ISO 13485 Lead Implementer training enables you to develop the necessary expertise to support an organization in establishing, implementing, managing and maintaining a Medical Devices Quality Management System (MDQMS) based on ISO 13485. During this training course, you will also gain a thorough understanding of the best practices of Medical Devices Quality Management Systems and be able to improve an organization`s overall performance by consistently providing safe and qualitative medical devices.
After mastering all the necessary concepts of Medical Devices Quality Management Systems, you can sit for the exam and apply for a “PECB Certified ISO 13485 Lead Implementer” credential. By holding a PECB Lead Implementer Certificate, you will be able to demonstrate that you have the practical knowledge and professional capabilities to implement ISO 13485 in an organization.
Who Should Attend?
Learning objectives
This training course will help you:
Educational approach
Prerequisites
A fundamental understanding of ISO 13485 and comprehensive knowledge of implementation principles.
Course Agenda
Day 1: Introduction to ISO 13485 and initiation of a MDQMS
Day 2: Plan the implementation of a MDQMS
Day 3: Implementation of a MDQMS
Day 4: MDQMS monitoring, measurement, continuous improvement and preparation for a certification audit
Day 5: Certification Exam
Examination
The “PECB Certified ISO 13485 Lead Implementer” exam fully meets the requirements of the PECB Examination and Certification Programme (ECP). The exam covers the following competency domains:
Domain 1: Fundamental principles and concepts of a Medical Devices Quality Management System (MDQMS)
Domain 2: Medical Devices Quality Management System (MDQMS)
Domain 3: Planning a MDQMS implementation based on ISO 13485
Domain 4: Implementing a MDQMS based on ISO 13485
Domain 5: Performance evaluation, monitoring and measurement of a MDQMS based on ISO 13485
Domain 6: Continual improvement of a MDQMS based on ISO 13485
Domain 7: Preparing for a MDQMS certification audit
General Information
1. Certification fees are included on the exam price.
2. Training material containing over 450 pages of information and practical examples will be distributed.
3. An attestation of course completion worth 31 CPD (Continuing Professional Development) credits will be issued to the participants who have attended the training course.
4. In case of exam failure, you can retake the exam within 12 months for free.
Certification
After successfully completing the exam, you can apply for the credentials shown on the table below. You will receive a certificate once you comply with all the requirements related to the selected credential.
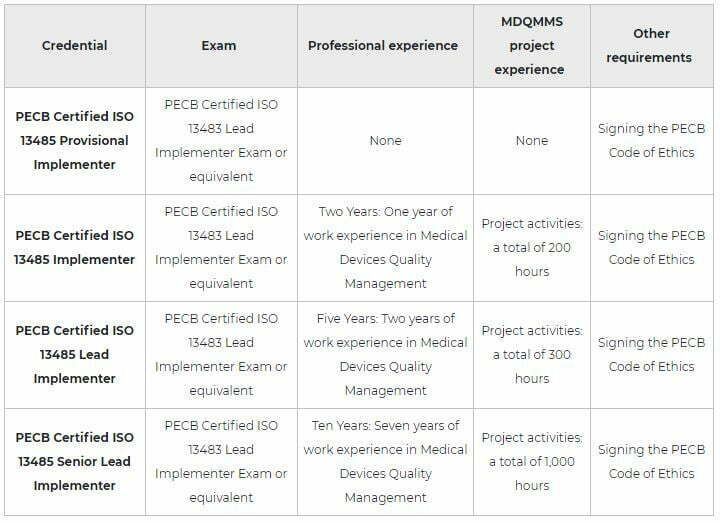
Note: PECB Certified Individuals who do possess the Lead Implementer and Lead Auditor Credentials are qualified for the respective PECB Master Credential, given they have taken 4 additional Foundation Exams which are related to this scheme. For more detailed information about the Foundation Exams and the overall Master Requirements, please go to the following link: https://pecb.com/en/master-credentials.
To be considered valid, these implementation activities should follow best implementation practices and include the following activities:
1. Drafting a MDQMS plan
2. Initiating a MDQMS implementation
3. Implementing a MDQMS
4. Monitoring and managing a MDQMS implementation
5. Performing continual improvement measures

